First in Florida to Offer New Glaucoma Treatment Procedure
The XEN Gel Stent is an exciting new procedure for glaucoma patients that is less invasive and highly effective. Our own Dr. Rajesh Shetty was selected to be the first doctor in Florida outside of the FDA study to perform this procedure.
Overview of Standard Glaucoma Treatments
Currently, patients who suffer from glaucoma are started out on a treatment of eye drops and medication, which is often followed by a something known as laser trabeculoplasty and in the worse case, a filtering procedure that lets the fluid drain outside of the eye.
The most common surgeries have used a drainage implant known as a trabeculectomy, and have been for several decades. While these have been effective, they can be more invasive and common complications with such procedures can be severe and vision-threatening.
In the world of minimally invasive glaucoma surgery (often called MIGS for short), there are some new devices that are helping to change the landscape and reduce the risks.
The XEN Gel Stent: New Glaucoma Treatment Now Available
All over the world, physicians agree that having the subconjunctival outflow pathway gives the greatest intraocular pressure (IOP) reduction for patients, which has been proven over and over again over the last 50 years. So the XEN Gel Stent is a solution to the need to have an outflow pathway, while reducing other risks and complications with the current procedures.
The XEN Gel Stent was developed by AqueSys and was later acquired by Allergan and was FDA approved in November of 2016 for the management of refractory glaucomas with those who haven’t seen success with surgical options or who had primary open angle glaucoma. It was also approved for those with pseudoexfoliative or pigmentary glaucoma with open angles that are not responding to the highest levels of medical therapy.
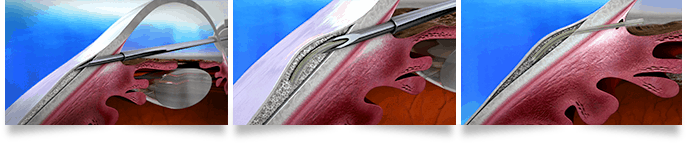
Made of a permanent and soft collagen-derived gelatin, the XEN Gel Stent allows for a minimally invasive and highly effective subconjunctival outflow pathway. After it is in place, it gently allows an outflow of aqueous from the anterior chamber into the non-dissected tissue of the subconjunctival space. Because it is flexible and soft, it conforms to the tissue around your eye, which may be shown to minimize many of the issues seen in other synthetic materials. It does not produce inflammation because the gelatin is well accepted by the human body.
This means that it lets the eye use a subconjunctival drainage pathway without the potential complications of a trabeculectomy. Best of all, the patient outcomes are supporting its continued use.
Benefits of XEN Gel Stent
This procedure is minimally invasive which reduces complications during the procedure, it is made of a soft gelatin which improves the body’s acceptance of it over other synthetic materials, and it has shown that it can significantly reduce IOP through the subconjunctival outflow pathway over a long period of time. Patients are often able to see very well the following day without much swelling at all.
It’s important to note that no treatment is guaranteed to completely eliminate the need for eye drops, but the strong results seen in the use of the XEN Gel Stent give the right patients a very strong choice for eliminating the need for them.
To talk to one of our highly trained physicians about the XEN Gel Stent, contact us today. Our mission is to provide the best eye care to our patients, using the safest and most advanced technology available.